 
The product was developed in Israel and is manufactured at Rafa’s site in Jerusalem. Rafa said FDA approval followed an expedited review. The DOD’s Chemical and Biological Defense Program also supported this effort. Rafa and DoD previously collaborated on the development of Rafa’s Atropine autoinjector which was launched in the U.S. This product could assist in saving many lives around the globe.” “Clinical studies confirmed the correlation between early treatment of status epilepticus and a reduced risk of ongoing and irreversible neurological damage. :max_bytes(150000):strip_icc()/midazolam-566f974d5f9b583dc3846b92.jpg) 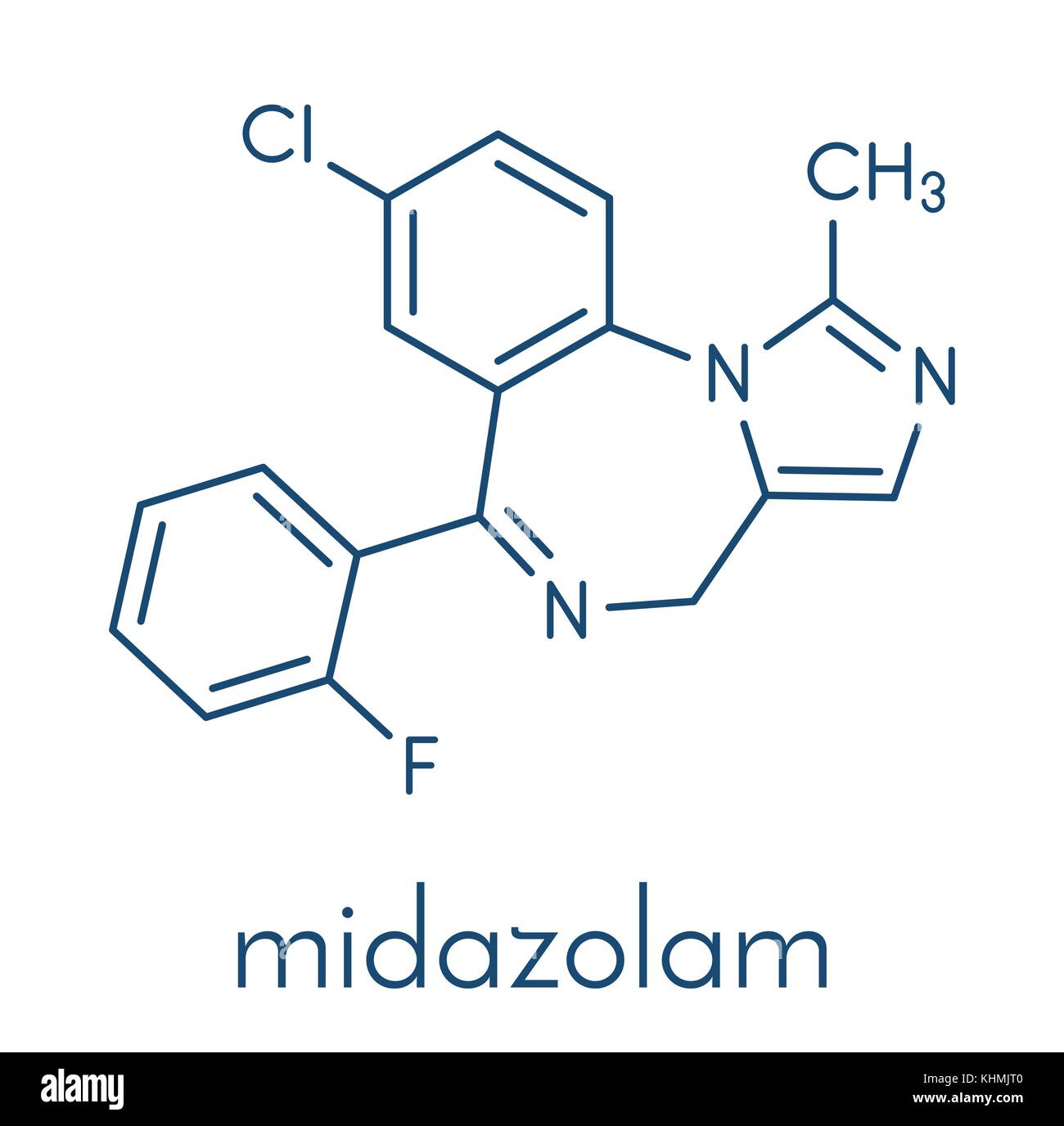
“The usability of the autoinjector has a significant medical advantage in its immediate treatment effect, as well as in reducing long-term damage,” Rafa Head of Emergency Solutions Roy Shay said in the release. These are significant advantages in a field setting during an emergency when fast treatment reduces the likelihood of permanent damage that could result from a continuous seizure, Rafa said in a news release. The autoinjector is administered intra-muscularly to the thigh in a simple and fast manner that does not require an intravenous line. 
JPEO-CBRND determined that the autoinjector improves upon and will replace the currently fielded convulsant antidote for nerve agent (CANA), the diazepam autoinjector. This means the new autoinjector can be used to treat seizures resulting from nerve agent exposure. Rafa’s Midazolam 10mg autoinjector received FDA indication for the treatment of status epilepticus in adults. Department of Defense’s (D0D) Joint Program Executive Office for Chemical, Biological, Radiological and Nuclear Defense (JPEO-CBRND). Jerusalem-based Rafa developed the autoinjector through cooperation with the U.S. Rafa Laboratories announced today that it received FDA approval for its 10mg Midazolam autoinjector for treating status epilepticus, or prolonged seizures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
